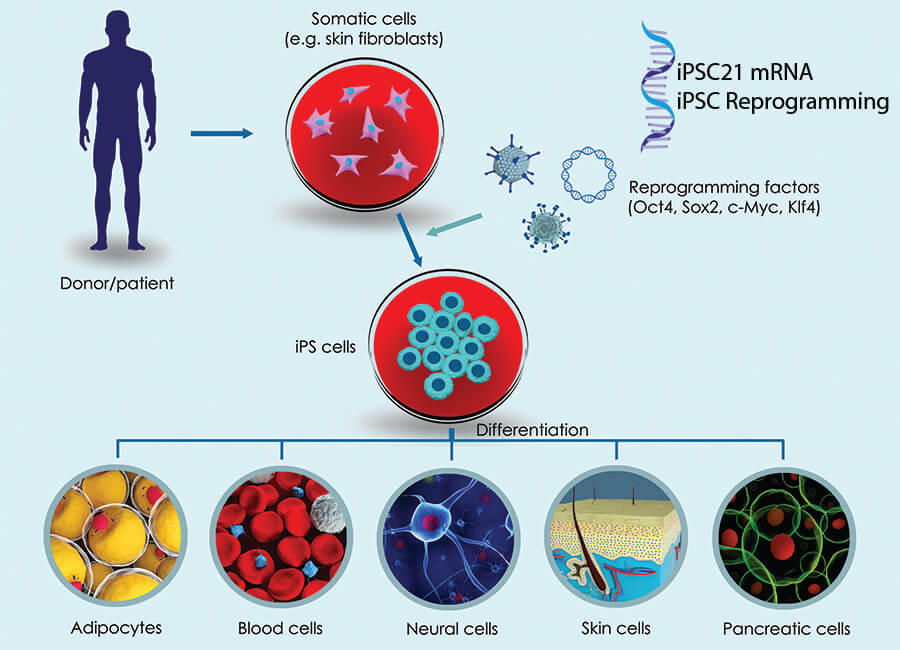
iPSC Reprogramming
The iPSC21 mRNA reprogramming method uses a transgene-free, footprint-free iPSC generation, with a non-integrating transfection system. This provides safety to the iPSC to be used in drug discovery and cell therapy applications. Molecular and functional assays such as flow cytometry and in-vitro embryoid body formation is used to characterize the pluripotency of the newly established iPSC cell line. Saftey tests (myoplasma, bacteriology, endotoxin and viral testing) and genetic integrity profiles are performed on the iPSC lines at point of cryogenic-freezing, and when cell therapies are developed.
mRNA iPSC Reprogramming
Our method of fibroblast reprogramming to Induced Pluripotent Stem Cells uses a custom mRNA method. Our protocol generates CLEAN iPSCs which can e used in the development of therapeutic applications.
The iPSC21 protocol requires on average 2 months to cultures the skin fibroblasts (starting cell type), reprogram the cells with our mRNA method, subculture and expand the generated iPSCs, then finally the validation of iPSCs (some parameters are shown below). At this stage, the iPSCs are cryogenically preserved for future use.
All iPSC21 customers will get a full certification of analysis (COA)
Generating Induced Pluripotent Stem Cells
iPSC Stages of Development
During the initial reprogramming phase where fibroblasts are transforming to iPSCs there is a very noticeable morphology change. This alteration in the cells appearance continues for around a 2 weeks at which point the iPSCs begin to proliferate.
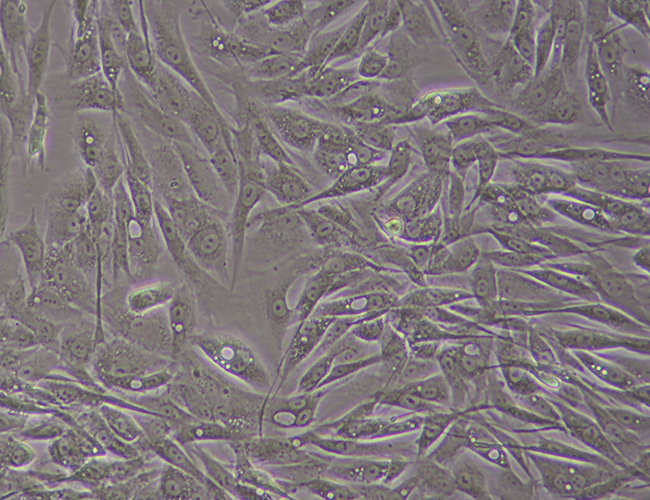
Skin-derived Fibroblasts
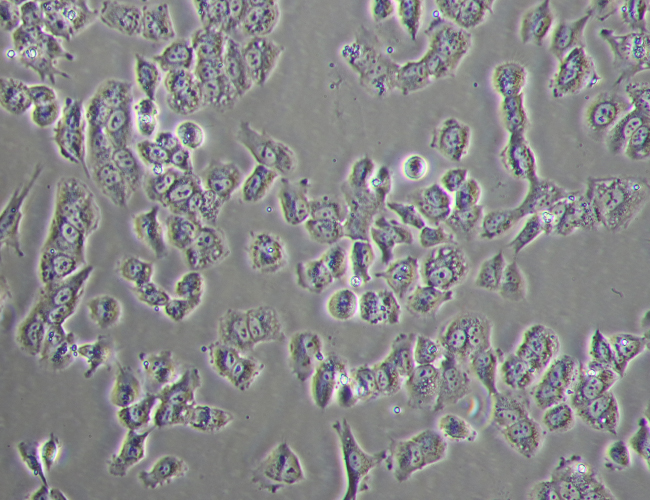
iPSC Reprogramming
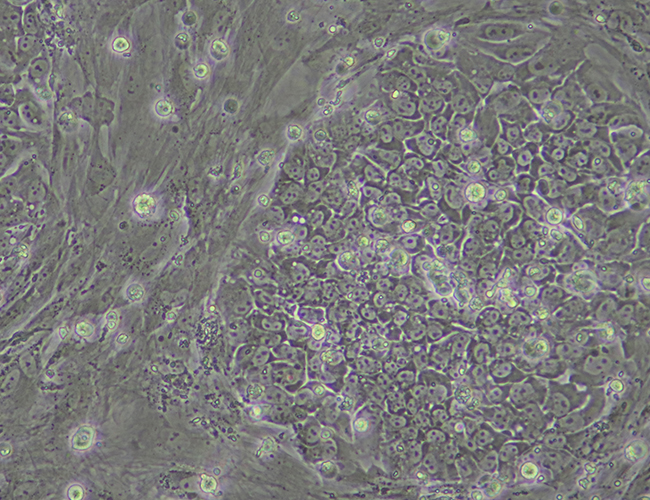
iPSC Colony
iPSC Subculture
At this stage the induced pluripotent stem cell colonies are selected and subcultured as pure iPSC lines. The pure cell lines are grown to expand colony size and cell numbers. During this period further steps are taken to develop stability in the newly establish iPSC lines.
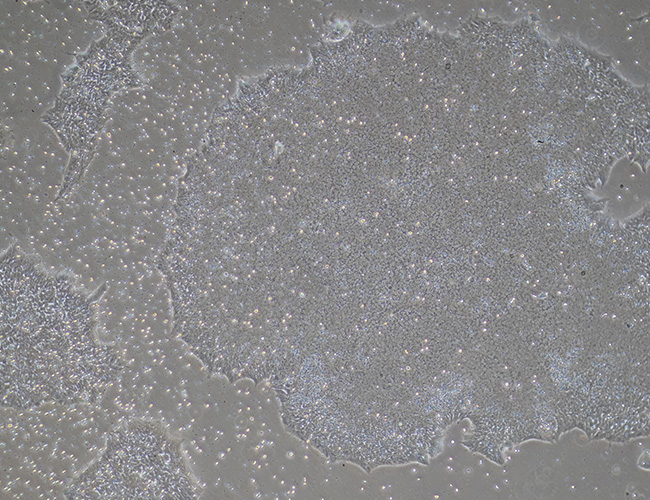
Expanding iPSCs

Pure iPSC Line

Stable iPSC Line
iPSC Characterisation
Immunocytochemistry Staining (ICC)
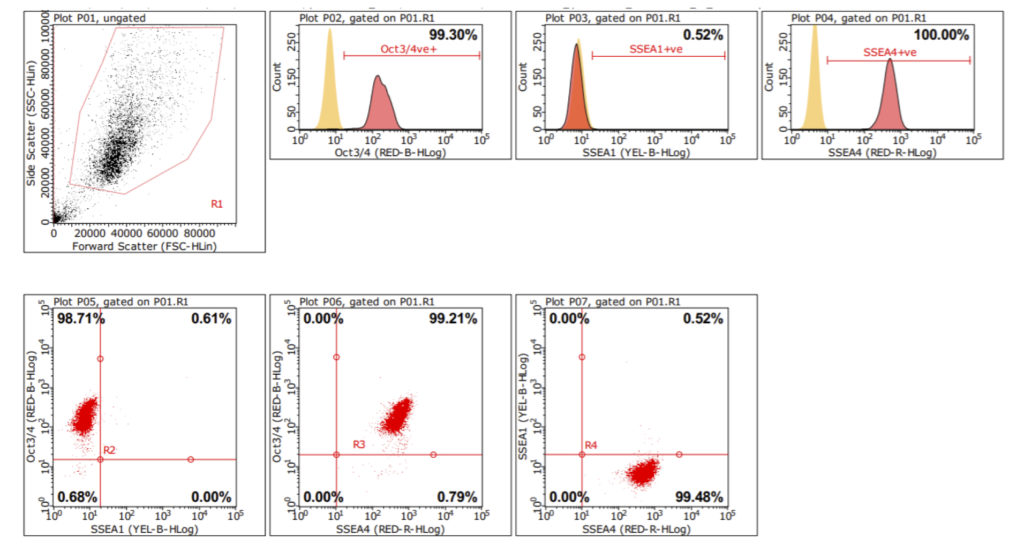
Alkaline phosphatase (AP) is a universal pluripotent marker for all types of pluripotent stem cells including embryonic stem cells, embryonic germ cells, and induced pluripotent stem cells. The pluripotent status of stem cells can be characterized by a high level of AP expression, along with the expression of multiple pluripotency markers including the transcription factors Nanog, Oct4, Sox2, stage-specific embryonic antigens, SSEA-1, -3, -4, and tumor-related antigens, TRA-1-60, TRA-1-81.
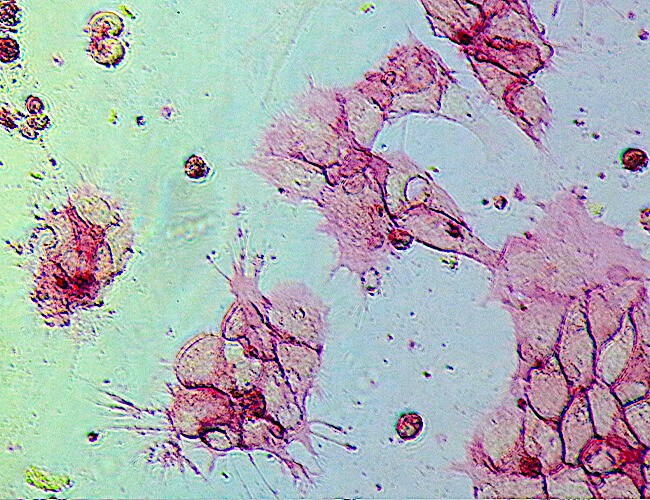
Alkaline Phosphatase (AP)

OCT-4
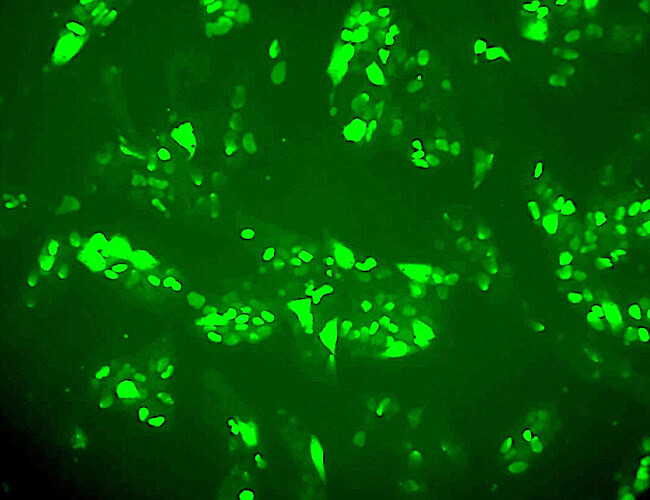
SOX-2
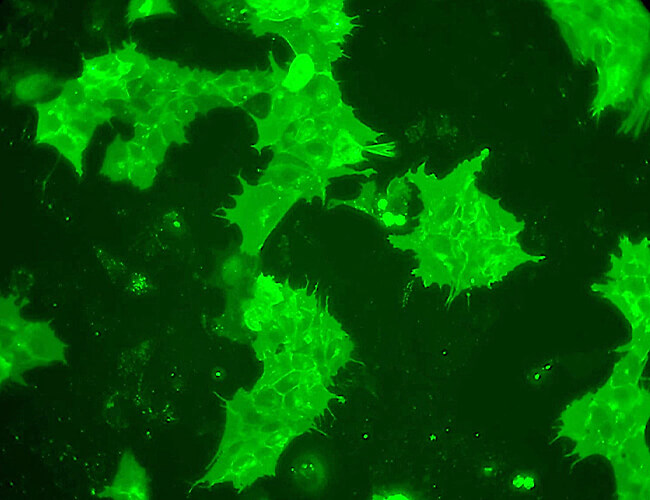
TRA-1-60

SSEA-4
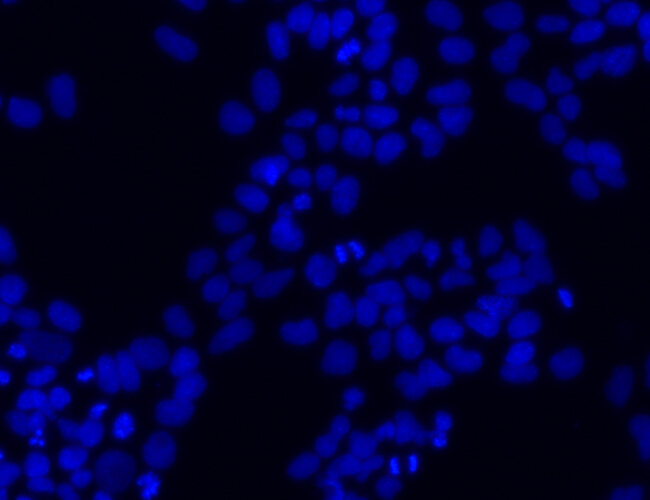
DAPI
iPSC Phenotype by Flow Cytometry
Flow cytometry allows for population characteristics to be assessed. Looking at the same key markers as for ICC (OCT4 and SSEA4) flow cytometry provides single-cell resolution allowing the purity of the line to be determined, as well as viability, cell count and the size and shape of the cells. All data was compared to isotype controls.